
Figure 4 from Structure and binding energy of the H2S dimer at the CCSD(T) complete basis set limit. | Semantic Scholar

Fragment molecular orbital (FMO) calculations on DNA by a scaled third-order Møller-Plesset perturbation (MP2.5) scheme - ScienceDirect

The calculated binding energies in cm 1 ) at the MP2 , MP3 , MP4 , CCSD... | Download Scientific Diagram
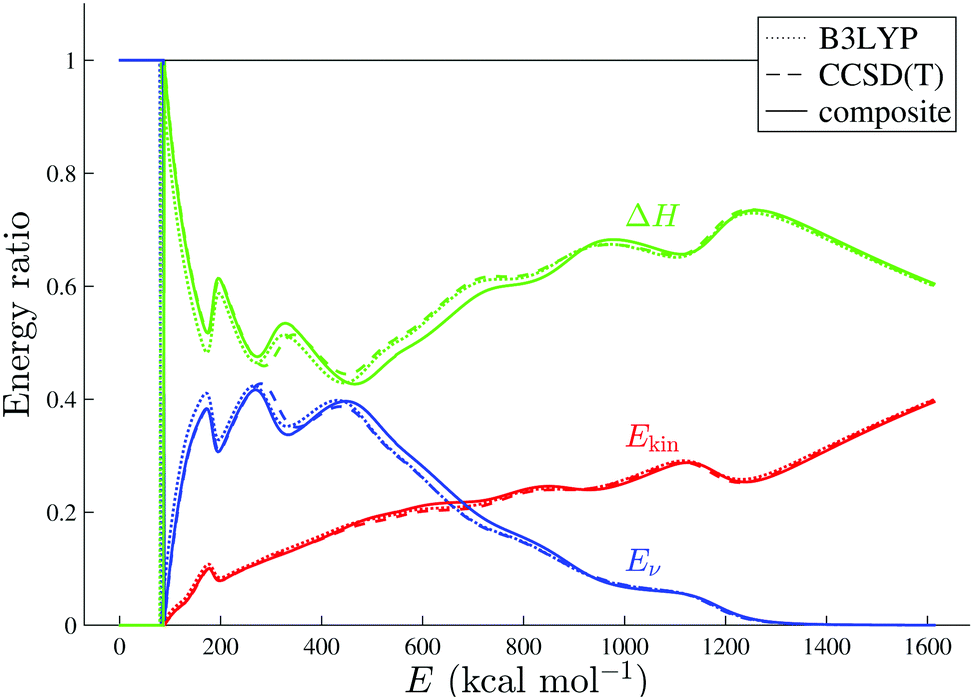
Statistical molecular fragmentation: which parameters influence the branching ratios? - Physical Chemistry Chemical Physics (RSC Publishing)

Pro®le diagrams of the di erence electron density distribution ¢»…r † ˆ... | Download Scientific Diagram

Efficient and accurate approximations to the local coupled cluster singles doubles method using a truncated pair natural orbital basis: The Journal of Chemical Physics: Vol 131, No 6

Theoretical insight into structural and electronic properties of cationic Scn+ (n=2-13): A benchmark study - ScienceDirect
![Molecules | Free Full-Text | Relationships between Interaction Energy and Electron Density Properties for Homo Halogen Bonds of the [(A)nY–X···X–Z(B)m] Type (X = Cl, Br, I) | HTML Molecules | Free Full-Text | Relationships between Interaction Energy and Electron Density Properties for Homo Halogen Bonds of the [(A)nY–X···X–Z(B)m] Type (X = Cl, Br, I) | HTML](https://www.mdpi.com/molecules/molecules-24-02733/article_deploy/html/images/molecules-24-02733-ag.png)
Molecules | Free Full-Text | Relationships between Interaction Energy and Electron Density Properties for Homo Halogen Bonds of the [(A)nY–X···X–Z(B)m] Type (X = Cl, Br, I) | HTML

Figure 2 from Structure and binding energy of the H2S dimer at the CCSD(T) complete basis set limit. | Semantic Scholar

Walltime for the CCSD(T) calculation of total energy of (H 2 O) 20 as a... | Download Scientific Diagram
The calculated binding energies in cm 1 ) at the MP2 , MP3 , MP4 , CCSD... | Download Scientific Diagram
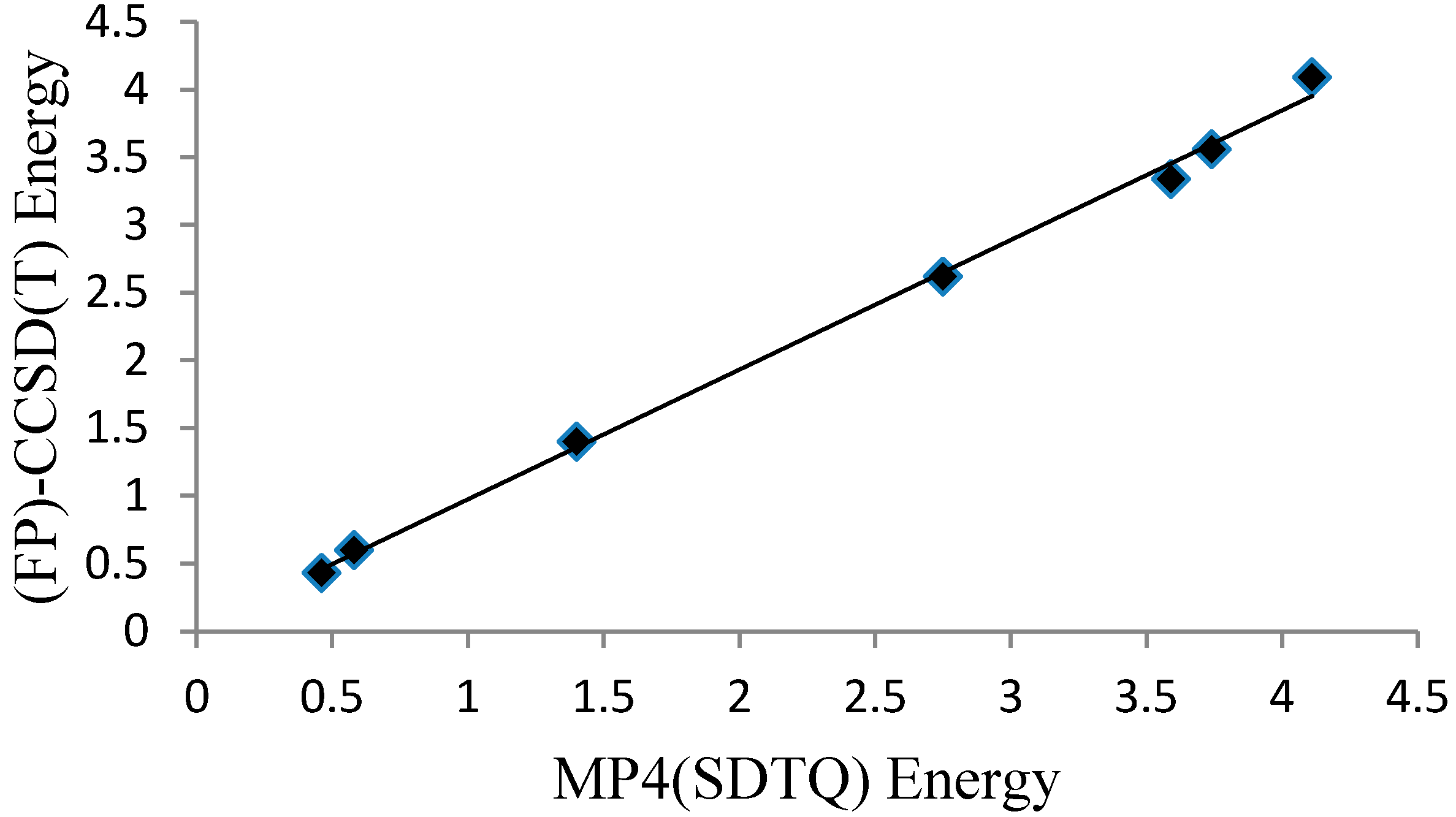
Molecules | Free Full-Text | Toward Exploring Novel Organic Materials: MP4-DFT Properties of 4-Amino-3-Iminoindene | HTML

On the applicability of the MP2.5 approximation for open-shell systems. Case study of atmospheric reactivity - ScienceDirect

Application of a convergent, composite coupled cluster approach to bound state, adiabatic electron affinities in atoms and small molecules: The Journal of Chemical Physics: Vol 144, No 1

Figure 3 from Structure and binding energy of the H2S dimer at the CCSD(T) complete basis set limit. | Semantic Scholar

Molecules | Free Full-Text | Toward Exploring Novel Organic Materials: MP4-DFT Properties of 4-Amino-3-Iminoindene | HTML

Benchmark theoretical study of the electric polarizabilities of naphthalene, anthracene, and tetracene: The Journal of Chemical Physics: Vol 138, No 2

Ab initio correlation functionals from second-order perturbation theory: The Journal of Chemical Physics: Vol 125, No 10

Recent progress on discovery and properties prediction of energy materials: Simple machine learning meets complex quantum chemistry - ScienceDirect
Full article: Electronic spectroscopy of some small anions containing S, N and O using CR-EOM-CCSD(T) method
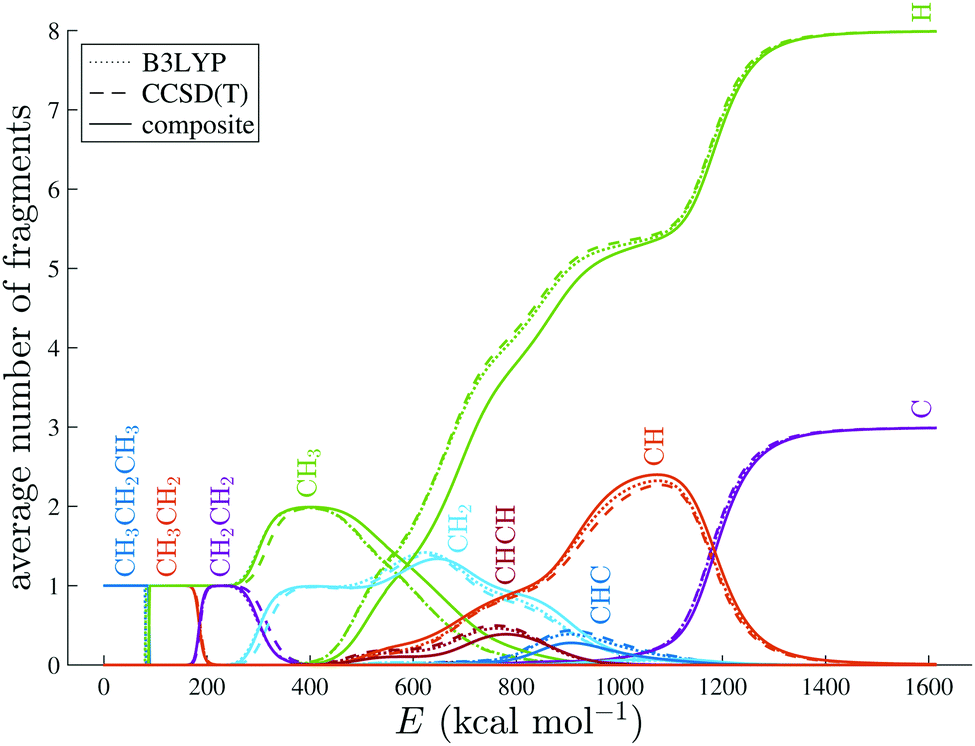
Statistical molecular fragmentation: which parameters influence the branching ratios? - Physical Chemistry Chemical Physics (RSC Publishing)

Levels of symmetry adapted perturbation theory (SAPT). I. Efficiency and performance for interaction energies: The Journal of Chemical Physics: Vol 140, No 9

The calculated binding energies in cm 1 ) at the MP2 , MP3 , MP4 , CCSD... | Download Scientific Diagram

On the applicability of the MP2.5 approximation for open-shell systems. Case study of atmospheric reactivity - ScienceDirect

Computerized implementation of higher‐order electron‐correlation methods and their linear‐scaling divide‐and‐conquer extensions - Nakano - 2017 - Journal of Computational Chemistry - Wiley Online Library

